Factsheets
Candidaemia
| NAMES Candidaemia (bloodstream infection), disseminated candidiasis, invasive candidiasis. For a recent review, see Kullberg & Arendrup (2015). |
| DISEASE Most (~90%) cases of systemic candidiasis arise from the gastrointestinal tract, and the remainder from venous catheters or other instrumentation. Candida bloodstream infections may be silent or associated with fever, reduced oxygenation, features of sepsis, and occasionally rash. If infection is silent, then organ involvement (usually liver, kidneys, spleen, occasionally brain, retina of the eye, bone or other organs) is usually not recognised unless a biopsy is taken, as clinical features are not specific. |
| FUNGI Candida albicans, Candida glabrata, Candida tropicalis, Candida parapsilosis, Candida krusei, Candida auris, other Candida spp. |
| GLOBAL BURDEN Candidaemia occurs at a population rate of 2-11/100,000, so ~350,000 cases are predicted worldwide every year. These numbers grew in the US by 52% between 2000 and 2005. Rates in India and Brazil are much higher, so the overall estimate could be greater. Blood cultures are insensitive, so the burden of invasive candidiasis is likely to be greater. |
| RISK FACTORS Prematurity, especially extremely low birth weight infants, chemotherapy and neutropaenia, critical care, pancreatitis (17.5% rate), major trauma, burns, multiple antibiotics, renal dialysis or dysfunction, central venous catheterisation. There is an increased risk of candidaemia associated with longer stays in intensive care, notably with COVID-19 patients. |
| DIAGNOSIS Blood culture. PCR test is more sensitive than blood culture for invasive candidiasis. Tissue biopsy. Candida antigen and antibody tests. Detailed examination of the retina (YouTube video Liu et al, 2015) |
| TREATMENT Caspofungin, micafungin, anidulafungin, amphotericin B (including lipid formulations), fluconazole and voriconazole. Flucytosine may have a role in endocarditis and endophthalmitis as well as urinary tract infections. Antifungal resistance is especially a problem with fluconazole. Resistance to echinocandins is not frequent but case reports and small series have been described. Current guidelines |
| OUTLOOK Candidaemia has a mortality of 30-55%. Invasive candidiasis is a serious problem with a high morbidity and mortality. |
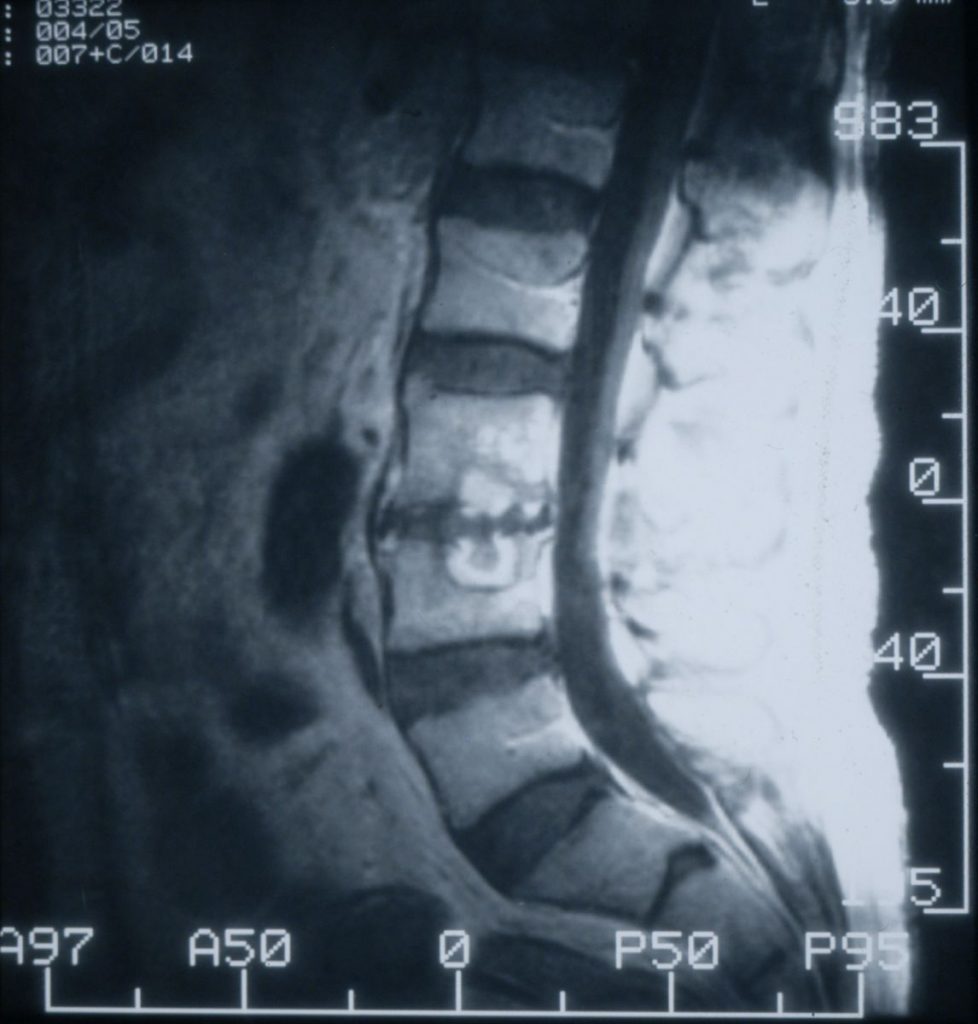
MRI of the lower lumbar spine and sacrum showing 2 affected vertebral bodies with a disrupted intervertebral disc, caused by Candida albicans (aspiration biopsy) 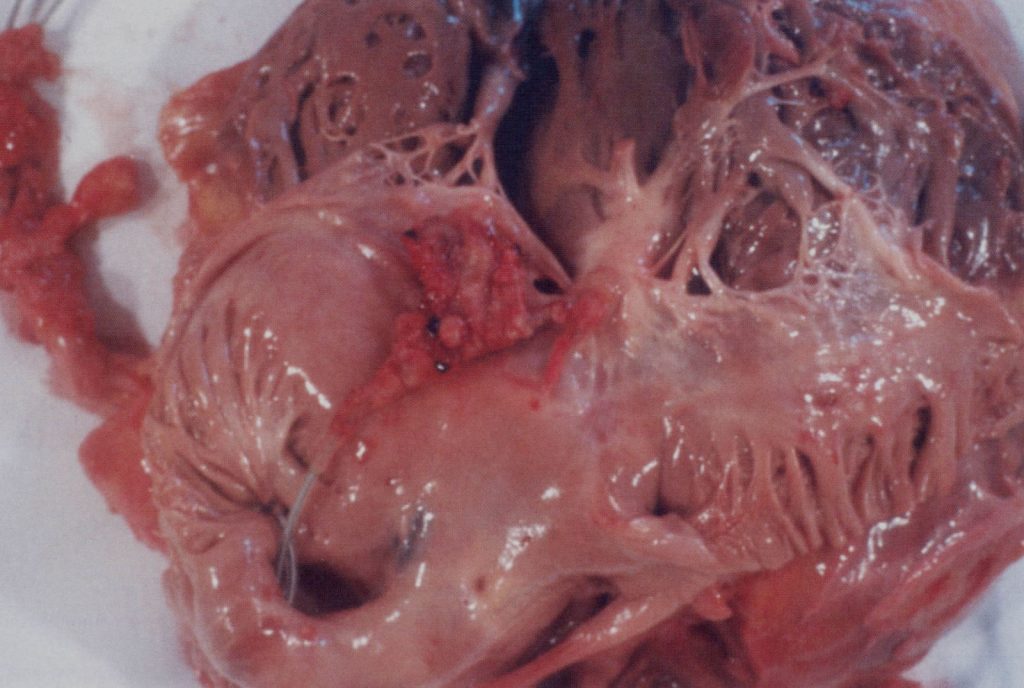
Candida pacemaker wire infection in the heart, after death 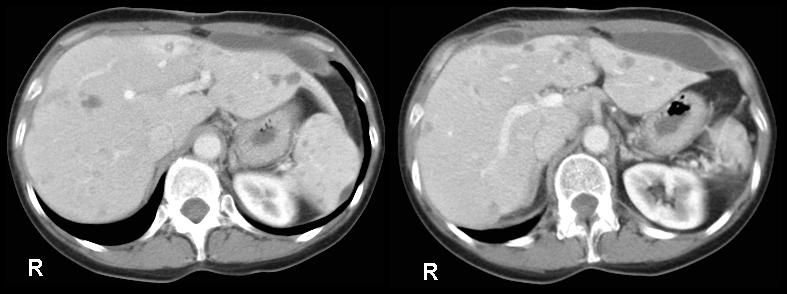
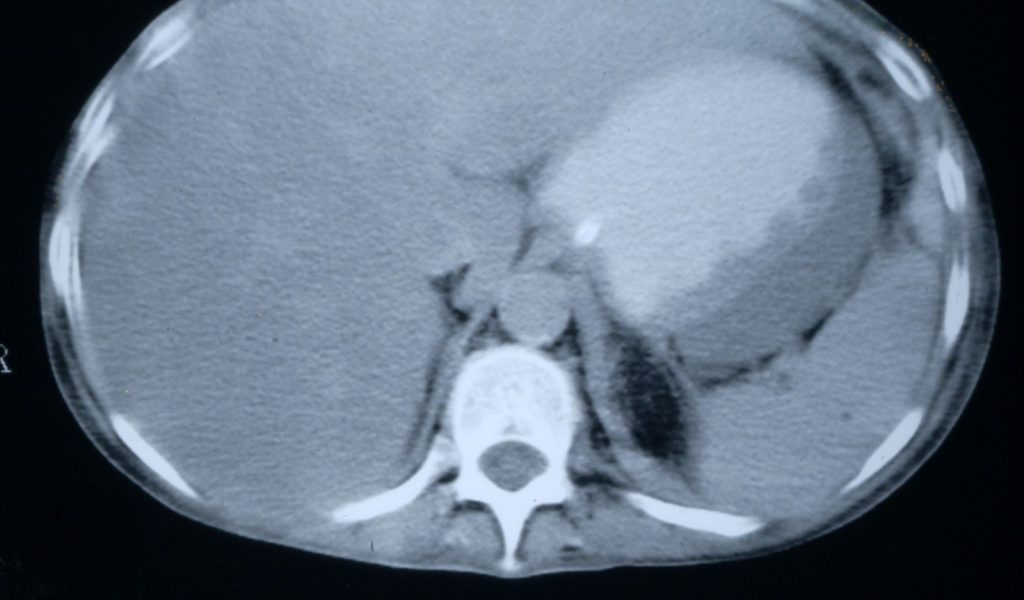
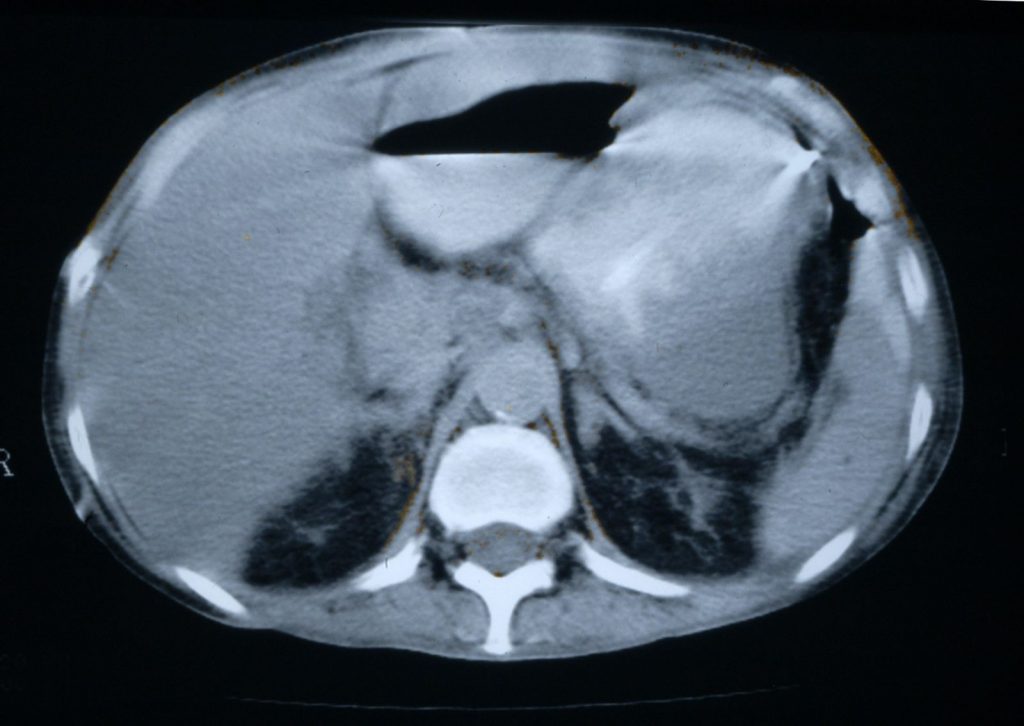
Disseminated candidiasis following an allogeneic stem cell transplant, showing multiple abscesses in the liver and spleen, fro 2 separate cuts of an abdominal CT scan. Aspiration of the small abscess anterior to the liver yielded Candida albicans DNA by PC 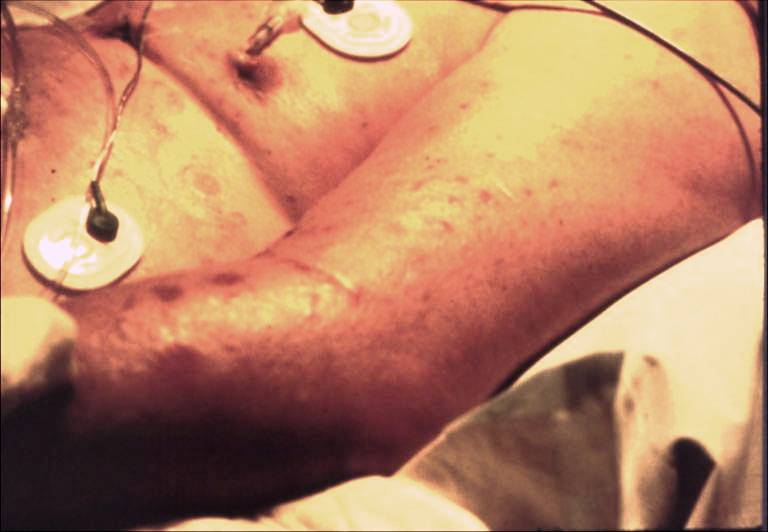
Rash caused by Candida tropicalis bloodstream infection in a cardiothoracic surgery patient 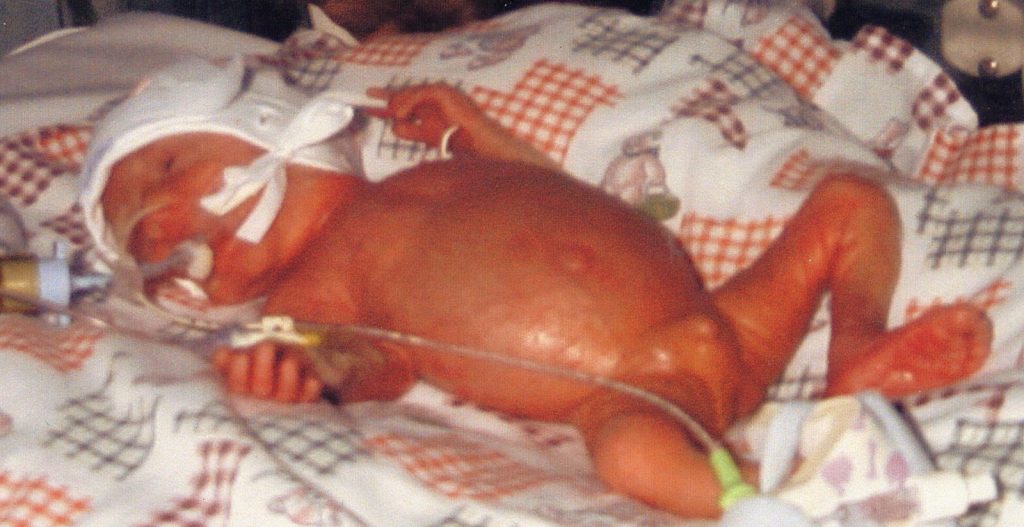
Premature infant who developed disseminated Candida infection who later died

Candida peritonitis
| NAMES Candida peritonitis, intra-abdominal candidiasis. For a recent review see Vergidis et al (2016) |
| DISEASE Follows abdominal surgery, pancreatitis or other intra-abdominal sepsis, or as a complication of peritoneal dialysis. The symptoms are indistinguishable from those of bacterial peritonitis, and bacterial and intra-abdominal candidiasis may occur together. It may present as tertiary peritonitis (persistent/recurrent infection following apparently adequate therapy of primary/secondary peritonitis), or as a localised intra-abdominal abscess. Abdominal discomfort, continuing purulent discharge from an abdominal drain, fever, raised inflammatory markers and white blood cell count are all common features, although often milder than bacterial peritonitis. In those receiving peritoneal dialysis, cloudy bags are typical and may be the first sign of infection. Necrotic pancreatitis is another common cause. |
| CLASSIFICATION – Primary peritonitis – Intra-abdominal abscess stemming from a GI tract peritoneal Candida infection from a intra-abdominal source – Secondary peritonitis stemming from a GI peritoneal Candida infection resulting from a intra-abdominal source – Intra-abdominal abscess stemming from a GI tract source – Secondary peritonitis stemming from a hepatobiliary or pancreatic source – Intra-abdominal abscess stemming from a hepatobiliary or pancreatic source – Infected pancreatic necrosis – Cholecystitis, cholangitis |
| FUNGI Candida spp., particularly C. albicans |
| GLOBAL BURDEN The frequency of post-surgical, intra-abdominal candidiasis is not known and is difficult to estimate. In a large multi-centre study of 101 French ICUs, hospital-acquired, intra-abdominal candidiasis was documented in 73 patients over eight months, compared with 123 patients with candidaemia without Candida peritonitis; 26 patients had both (Montravers et al, 2010). See also Vergidis et al, 2016. Assuming this can be generalised to other populations, this suggests a ratio of one patient with hospital-acquired (almost all post-operative) Candida peritonitis for every two patients with candidaemia, in ICU. As 30-50% of candidaemia cases occur in ICU, and there are about 300,000 episodes of candidaemia globally, this suggests about 50,000-75,000 cases of Candida peritonitis each year. Around 50% of patients with end stage renal disease (~1.7 million worldwide) receive chronic ambulatory peritoneal dialysis (CAPD). On average, patients get an infection every 18 months, and ~0.05 episodes per patient year are attributable to Candida spp., equivalent to ~42,500 cases annually. |
| RISK FACTORS Post-surgery, Candida peritonitis is most common if the bowel has been perforated, especially following faecal peritonitis, or following anastomotic leakage. Other risk factors include multiple antibiotics and diabetes mellitus. In CAPD peritonitis, patients with Candida peritonitis had been on peritoneal dialysis for longer, usually had prior episodes of bacterial peritonitis for which they were treated with antibacterial therapy. |
| DIAGNOSIS In surgical patients, diagnosis is by microscopic examination or culture of the peritoneal fluid collected during the operation, or sterile aspiration. Candida peritonitis should be suspected if Candida is grown from a drain in situ. Some patients have candidaemia at the same time. About 65% of patients with Candida peritonitis also grow bacteria from their peritoneal or abscess fluid. Usually a single species of Candida is grown, but sometimes more than one; C. albicans is found in ~55% of cases. In CAPD Candida peritonitis, Candida alone is usually cultured from the cloudy bag fluid, with many different species cultured. Candida PCR on blood or peritoneal fluid and/or serum beta-D-glucan detection is likely to be diagnostic, but has not been well studied. |
| TREATMENT In post-surgical Candida peritonitis, drainage of any abscess, either surgically or percutaneously, is important. Antifungal therapy with an echinocandin (caspofungin, micafungin or anidulafungin) or, if the causative organism is susceptible, fluconazole is critical. In a recent French study, 20% of the isolates were fluconazole resistant. As many patients also have concomitant bacterial peritonitis, this should also be treated. In CAPD Candida peritonitis, a key success factor is early removal of the peritoneal catheter and return of the patient to haemodialysis. Fluconazole, an echinocandin or intravenous amphotericin B (sometimes with flucytosine) are the best treatment options. Prophylaxis with fluconazole reduced the occurrence of Candida peritonitis in at-risk surgical patients in RCTs (Eggimann et al, 1999). Current guidelines |
| OUTLOOK In the 1990s, the attributable mortality of post-surgical Candida peritonitis was ~37% (with ~57% overall mortality), but a recent French study indicates that the overall mortality has fallen to ~40%. In CAPD Candida peritonitis, early (24h) peritoneal catheter removal reduces mortality from 32% to 13%. |
Large retrogastric abscess attached to the posterior gastric wall of the stomach caused by Candida glabrata. The patient had had several prior laparotomies and attacks of intra-abdominal sepsis and prior duodenojejenual anastomosis, with complicating uretero-enteric fistula. Two years this was repaired, but a month post-operatively she became septic. Blood cultures grew pseudomonas bacteria. A CT scan showed this collection (9 x 2.5cm) behind the stomach and another in the pelvis (6cm). The percutaneous drain inserted into the abscess grew only Candida glabrata.