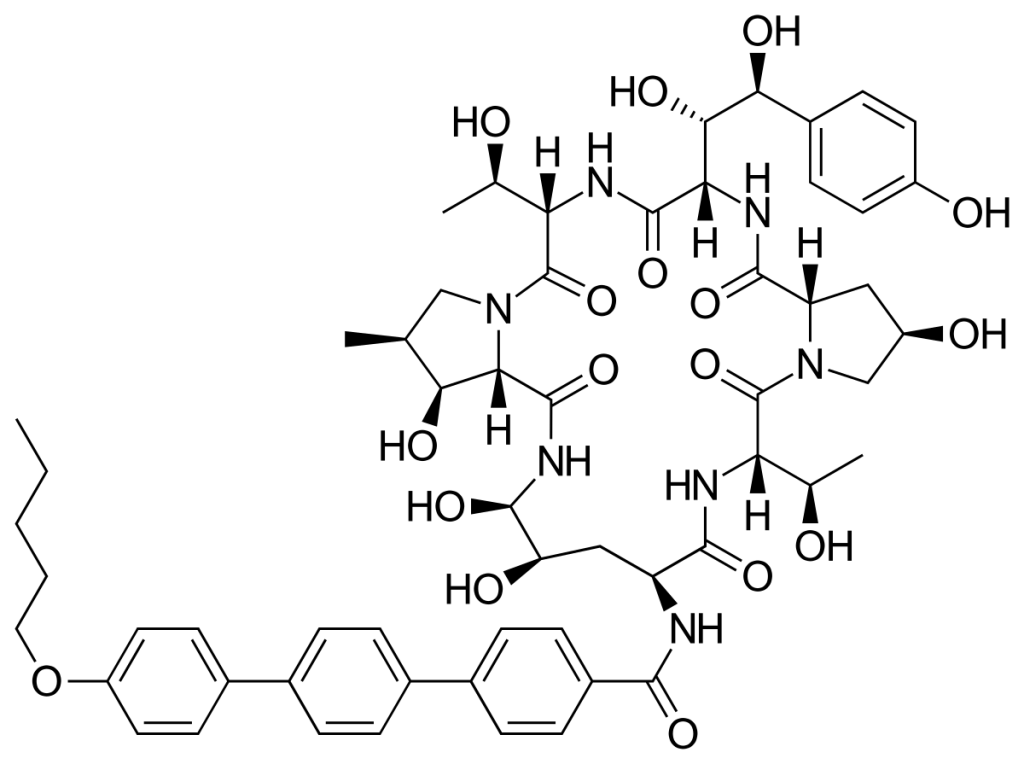
Echinocandins are a class of antifungal drug that inhibit the synthesis of β-D-glucan in the fungal cell wall via non-competitive inhibition of the enzyme 1,3 β-D-glucan synthase. These treatments are delivered intravenously.
Resistance to these drugs has been reported, particularly associated with mutations in the FKS gene (Hori & Shibuya, 2018). Due to a naturally-occurring variant of FKS in Candida parapsilosis, this strain exhibits a far greater resistance than other Candida species (Arendrup & Perlin, 2014).
Rezafungin (formerly known as CD101) is a new echinocandin currently in development. This drug has increased pharmacokinetic stability compared to previous formulations. Results of the Phase 2 STRIVE trial suggest it is effective for candidaemia and/or invasive candidiasis (Thompson et al, 2020). Two Phase 3 clinical trials are currently underway to determine its efficacy and safety for treatment (ReSTORE) and prevention (ReSPECT) of candidaemia and/or invasive candidiasis. For more information please refer to this review by Zhao & Perlin, 2020.

Videos

Factsheets
Caspofungin
| NAMES Caspofungin. Cancidas (Merck) and generic products For a review please see Hashemian et al (2020). |
| STRUCTURE & MECHANISM Semisynthetic derivative of a fermentation product of the fungus Glarea lozoyensis. |
| FORMULATIONS & DOSAGES IV only. The standard dose is a 70 mg loading dose, with subsequent daily doses of 50 mg. In patients weighing more than 80 kg it is recommended (in Europe) that 70 mg a day is given rather than 50 mg a day. One study showed that doses up to 200 mg/day are well tolerated (Cornely et al, 2011). Patients with moderate liver insufficiency should receive a dose of 35 mg a day and it is possible that the drug is not appropriate for patients with severe liver disease although this has not been decided. The appropriate dose in children is 50 mg/m2 daily. |
| ACTIVITY – ACTIVE against all species of Candida, with few exceptions. It is extremely active and usually rapidly fungicidal against all species except Candida parapsilosis, Candida guilliermondii and Candida famata, against which it is moderately active. It has activity against Candida biofilms, which azoles in general do not. Some resistant C. albicans, C. glabrata, C. tropicalis and C. krusei isolates have been described, almost all with mutations in two hot spots in the target gene (Perlin, 2015). Caspofungin is also active (although not fungicidal) against all Aspergillus species. It is not antagonistic when given in combination with amphotericin B or azoles against Aspergillus. – LIMITED ACTIVITY against Coccidioides spp., Blastomyces dermatitidis, Scedosporium spp., Paecilomyces varioti and Histoplasma spp. which is not sufficient for clinical use. Recent model data suggests possible synergistic activity with amphotericin B against Rhizopus oryzae. |
| TYPICAL REGIMEN For all indications, a 70 mg loading dose, then 50 mg a day (or 70 mg daily for patients weighing over 80 kg). It is possible that higher doses may be useful for some sanctuary site infections (brain, eye etc) and for isolates to which caspofungin has marginal activity, such as C. parapsilosis. The duration of therapy is uncertain but typically should be at least two weeks beyond the last evidence of active disease unless a switch to another antifungal is made. |
| PHARMACOKINETICS The drug is chemically degraded slowly in the body, probably in multiple organs including the liver. As a result there are very few alterations to be made in patients with organ dysfunction or taking other drugs, with the one exception of severe liver dysfunction. |
| DRUG INTERACTIONS Caspofungin is not an inhibitor and is a poor substrate for cytochrome people P450 enzymes, so drug interactions are few. It is not a substrate for P-glycoprotein. Concomitant use of ciclosporin with caspofungin increases the exposure to caspofungin by about a third. Caspofungin reduces the trough concentration of tacrolimus by about a quarter so in both instances monitoring (TDM) of these drugs is appropriate. Concomitant use of caspofungin with rifampicin increases caspofungin exposure but this changes over the first two weeks of therapy. Caspofungin dose should be increased to 70 mg with rifampicin, efavirenz, nevirapine, dexamethasome, phenytoin or carbamazepine. Tacrolimus levels should be monitored. |
| SIDE EFFECTS & TOXICITY Phlebitis was the commonest side effect in patients receiving caspofungin through a peripheral intravenous catheter. Fever, chills, abdominal pain, nausea, diarrhoea and vomiting were all common together with headache. All were mild and/or reversible when the drug was stopped. Raised liver enzymes occur in about 1 in 20 patients. Other side effects appeared to be rare. |
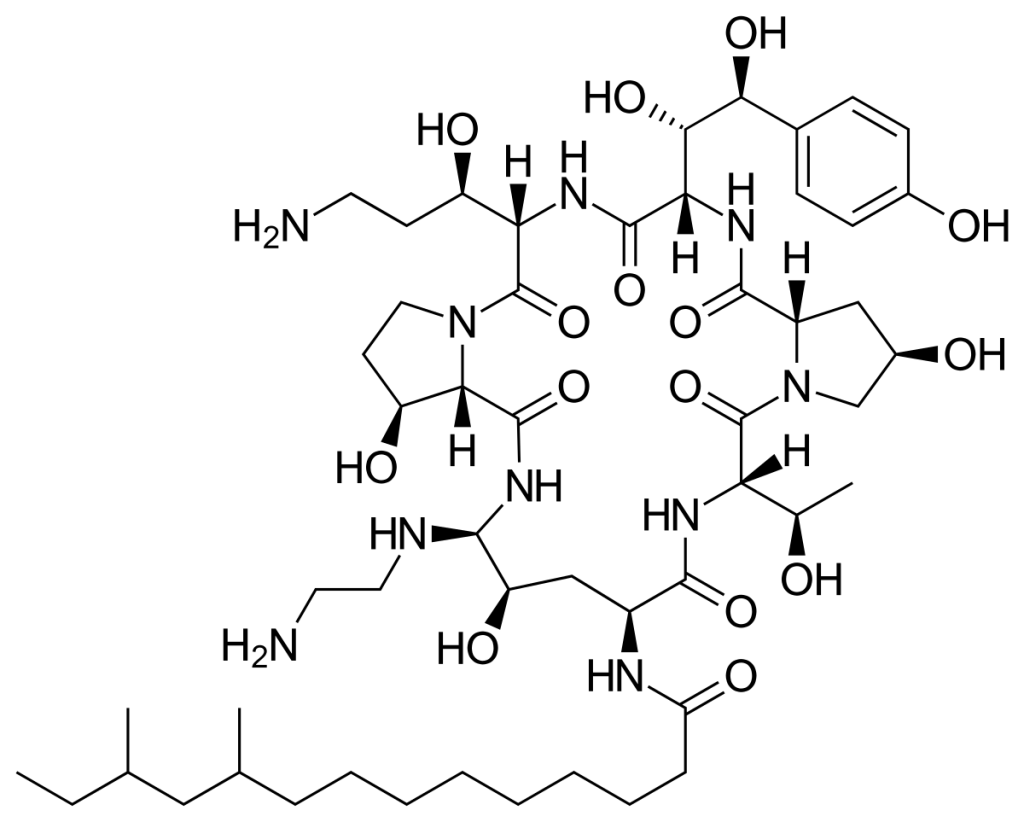

Micafungin
| NAMES Micafungin. Mycamine (Astellas) and generic products |
| APPROVAL 2005 (USA), 2002 (Japan), 2008 (Europe) |
| STRUCTURE & MECHANISM It is a semi-synthetic modification of a fermentation product from the fungus Coleophoma empetri, the lead compound first reported in 1990. It inhibits the function of beta-(1,3)-D-glucan synthase. |
| FORMULATIONS & DOSAGES IV only. The standard dose is 100 mg, but doses up to 8 mg/kg have been given safely. |
| ACTIVITY ACTIVE against all species of Candida. It is extremely active and usually rapidly fungicidal against all species except Candida parapsilosis, Candida guilliermondii and Candida famata against which it is moderately active. Its in vitro activity appears to be greater than other echinocandins, but may be counterbalanced by its higher protein binding. It has activity against Candida biofilms, which azoles in general do not. Some resistant C. albicans, C. glabrata, C. tropicalis and C. krusei isolates have been described, almost all with mutations in two hot spots in the target gene. Also active (but not fungicidal) against all Aspergillus species. It is not antagonistic when given in combination with amphotericin B or azoles against Aspergillus. |
| TYPICAL REGIMEN For invasive or chronic aspergillosis, at least 150 mg daily, for at least two weeks. Patients with mild or moderate liver insufficiency should receive the standard dose, but it is possible that the drug is not appropriate for patients with severe liver disease. The appropriate dose in children is 2 mg/kg daily and 10 mg/kg in premature infants. – For candidaemia, 100 mg daily (or 150 mg if neutropenic), for at least two weeks, or longer if candidaemia is slow to resolve. – For oesophageal candidiasis, 150 mg daily, usually for 5-7 days. – For invasive aspergillosis, at least 150 mg daily, for at least 2 weeks. The duration of therapy is uncertain but typically should be at least two weeks beyond the last evidence of active disease unless a switch to another antifungal is made. |
| PHARMACOKINETICS Reviewed by Wasmann et al (2018). The drug is metabolised in the body to several compounds (M1, M2, M5) some of which circulate. Exposure to these metabolites is low and they have no antifungal activity. No dose alterations need to be made in patients with organ dysfunction or taking other drugs, with the one exception of severe liver dysfunction. Rapid clearance in premature infants (gestational age ~26 weeks) is approximately 5-fold greater than in adults, so much greater doses are required. |
| DRUG INTERACTIONS Micafungin has few interactions with cytochrome people P450 enzymes, so drug interactions are few. It is not a substrate for P-glycoprotein. Micafungin is highly bound to plasma protein (> 99%), more than any other echinocandin. Micafungin may increase the exposure to sirolimus, nifedipine or itraconazole. Micafungin increases amphotericin B exposure by ~30%. |
| SIDE EFFECTS & TOXICITY Micafungin is well tolerated with occasional fever, phlebitis, headache, abdominal pain, nausea, diarrhoea, vomiting and rash. All were mild and/or reversible when the drug was stopped. Raised liver enzymes occur in up to 1 in 20 patients. Other side effects appeared to be rare. |
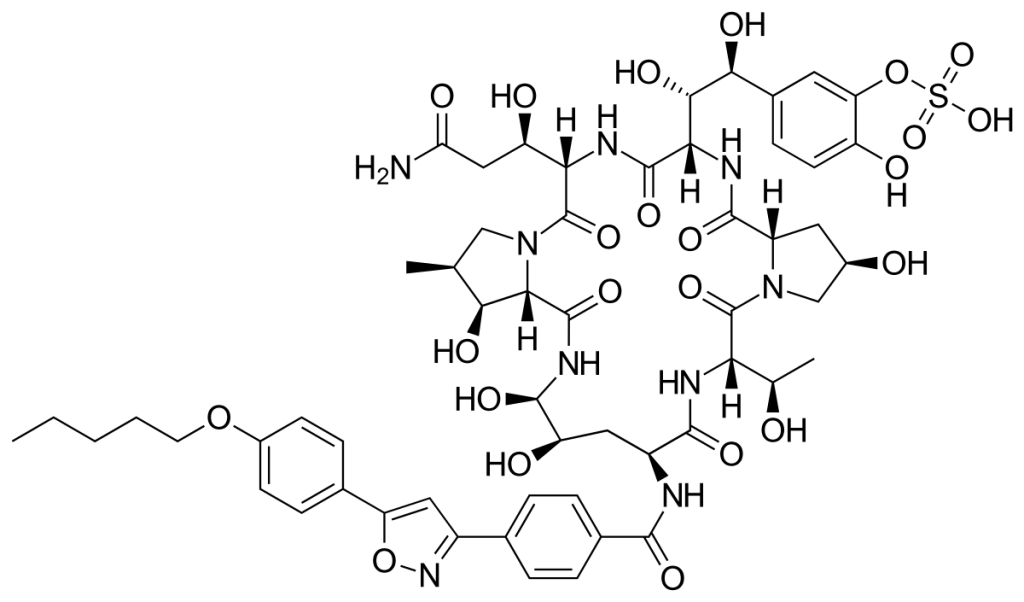

Anidulafungin
| NAMES Anidulafungin. Eraxis/Ecalta (Pfizer) |
| APPROVAL 2006 |
| STRUCTURE & MECHANISM It is a semi-synthetic modification of a fermentation product from the fungus Aspergillus nidulans. The lead compound for anidulafungin (LY303366) was identified in 1974. Numerous modifications failed to generate an orally bioavailable analogue and so Lilly out-licensed the product to Versicor (then Vicuron) in June 1999. Clinical studies commenced in 1998, but it was not licensed until 2006. It inhibits the function of beta-(1,3)-D-glucan synthase. |
| FORMULATIONS & DOSAGES IV only. For candidaemia and invasive candidiasis. The standard dose is a 200 mg loading dose, followed by 100 mg daily. No dosage adjustments are required for those with renal or hepatic insufficiency. Very few data support treatment in children. |
| ACTIVITY Anidulafungin is active against all species of Candida. It is extremely active and usually rapidly fungicidal against all species except Candida parapsilosis, Candida guilliermondii and Candida famata against which it is moderately active. It has potent activity against Candida biofilms, which azoles in general do not. Resistant C. glabrata isolates are recognised, some of which may retain susceptibility to other echinocandins. Resistance in C. albicans, C. tropicalis and C. krusei isolates have also been described usually confirmed by mutations in two hot spots in the target gene. Anidulafungin is also active (but not fungicidal) against all Aspergillus species. It is not antagonistic when given in combination with amphotericin B or azoles against Aspergillus. Resistance in A. fumigatus is described with anidulafungin and all the echinocandins, but difficult to measure with confidence. Anidulafungin has limited activity against Coccidioides spp., Blastomyces dermatitidis, Scedosporium spp., Paecilomyces varioti and Histoplasma spp. which is not sufficient for clinical use. |
| TYPICAL REGIMEN For candidaemia: 200 mg loading dose, then 100 mg daily. A maximum duration of therapy of 35 days is recommend, based on current preclinical exposure data. |
| PHARMACOKINETICS The drug is chemically degraded slowly in the body and has a large volume of distribution (30-50 L). Like other echinocandins, anidulafungin is extensively bound (>99%) to human plasma proteins. |
| DRUG INTERACTIONS Anidulafungin is not an inhibitor and is a poor substrate for cytochrome people P450 enzymes, so no drug interactions are documented. |
| SIDE EFFECTS & TOXICITY The commonest side effects (all less than 1 in 40 patients) were flushing or a hot flush, pruritus, rash and/or urticaria. Raised liver enzymes occur in about 1 in 40 patients. Other side effects appear to be rare. |