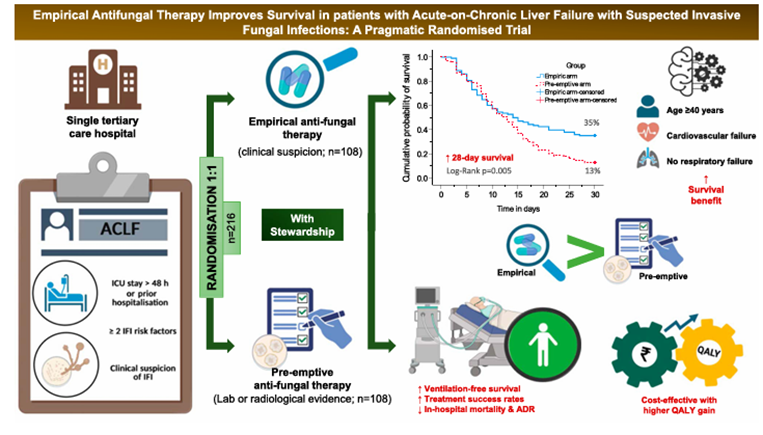
A randomised controlled trial conducted in India evidently showed that early empirical antifungal therapy, compared with pre-emptive therapy, improves survival in patients with acute-on-chronic liver failure and invasive fungal infections (IFIs). The significant findings were a higher treatment success and resolution of IFIs, and a lower in-hospital and IFI-attributable mortality in the empirical group (55.6% vs 75.9%; P = 0.003). In addition, fewer adverse events and an increased cost-effectiveness were observed.
The authors explained these observations as being linked to avoiding treatment delays, which were 4 days in the empirical group and 7.5 days in the pre-emptive group. Another factor was the timely initiation and the adequacy of antifungal treatment duration (typically at least 7–14 days), which is essential for translating antifungal activity into a measurable survival advantage.
Although the findings from this clinical trial were previously corroborated in other studies, such as Kollef et al, Garey et al., and Morrell et al, an earlier study had reported contrasting findings, stating pre-emptive antifungal therapy as not being inferior to empirical therapy in haematology patients. While this affirms the need for mycological, laboratory, or radiological evidence as essentials for informing stewardship decisions and stopping antifungals in patients without any evidence of IFI, a delayed turnaround time, particularly as seen in cultures, may negatively impact survival outcomes.